Exposure to fungal metabolites known as mycotoxins is increasingly recognized as a potential contributor to chronic health concerns in susceptible individuals. These compounds—produced by species of Aspergillus, Penicillium, Fusarium, and other fungal molds—can enter the body through contaminated food, indoor air exposure, or water-damaged environments. Once absorbed, mycotoxins may circulate through the bloodstream and undergo hepatic biotransformation before being eliminated through bile, urine, or stool.
In clinical practice, binder-based strategies are sometimes used as part of a broader approach to supporting mycotoxin elimination. Binders are substances that may adsorb certain toxins within the gastrointestinal tract, potentially reducing reabsorption during enterohepatic recirculation and promoting fecal excretion.
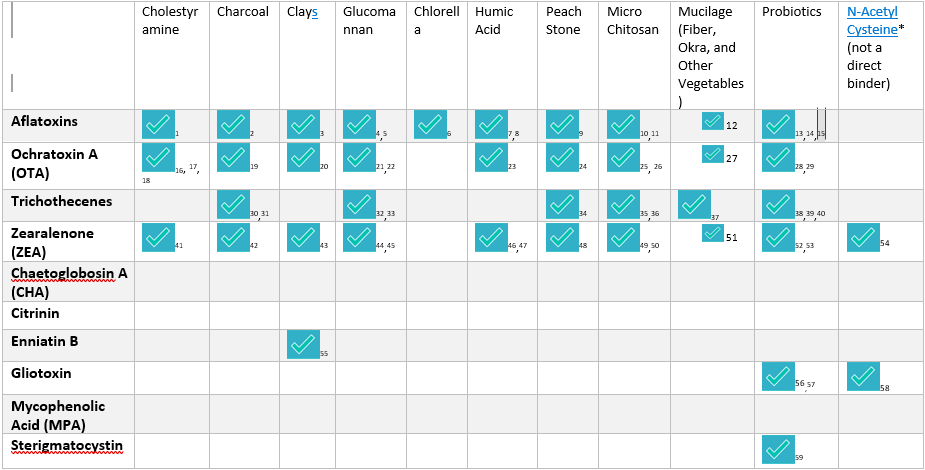
While the evidence base varies depending on the compound and toxin, multiple natural and pharmaceutical binders have demonstrated the ability to interact with specific mycotoxins in in vitro studies, animal models, or clinical observations.
Laboratory tools such as the Mosaic Diagnostics MycoTOX Profile can help clinicians identify patterns of mycotoxin exposure and guide personalized support strategies.
How Binders Work
A key part of understanding mycotoxins is knowing that many mycotoxins undergo enterohepatic circulation, meaning they are metabolized in the liver, secreted into bile, released into the intestines, and potentially reabsorbed into the bloodstream.
Binders can help mitigate this and function by:
- Adsorption – physically binding toxins through electrostatic attraction or surface adsorption
- Chelation or complex formation – forming stable complexes that limit absorption
- Interrupting enterohepatic recirculation – promoting elimination through stool
Different binders vary in their binding affinity for specific mycotoxins, which is why clinicians often use a combination approach depending on the toxin profile.
Cholestyramine
Cholestyramine is a bile acid sequestrant medication traditionally used to lower cholesterol levels. Because it binds bile acids in the intestine, it can also interrupt enterohepatic recirculation of certain toxins.
Research suggests cholestyramine may help bind lipophilic toxins, including some mycotoxins such as ochratoxin A and certain trichothecenes. By sequestering bile acids that contain toxin conjugates, cholestyramine may help reduce reabsorption and promote elimination through stool.
Clinically, cholestyramine is sometimes used in mold-related illness protocols due to its strong binding capacity, although it must be prescribed and monitored by a healthcare professional because it may also bind medications and nutrients.
Activated Charcoal
Activated charcoal is a highly porous carbon material found in all kinds of personal care products these days but is produced through controlled heating processes that create a large adsorption surface area.
Because of this structure, charcoal can bind a wide variety of compounds, including some mycotoxins. Studies suggest activated charcoal may adsorb:
- Aflatoxins
- Trichothecenes
- Ochratoxin A
Activated charcoal is widely used in toxicology for acute poisoning due to its ability to adsorb organic toxins within the gastrointestinal tract. In integrative clinical practice, it is sometimes used intermittently to support toxin elimination.
However, because charcoal can bind medications and nutrients, appropriate timing relative to supplements or pharmaceuticals is important.
Clay-Based Binders (Bentonite and Other Clays)
Natural clays such as bentonite, montmorillonite, and other aluminosilicates have been extensively studied for their ability to bind mycotoxins in agricultural and veterinary settings.
Clay particles carry a negative surface charge, allowing them to bind positively charged toxins through electrostatic interactions. This mechanism has been particularly well studied for:
- Aflatoxins
- Zearalenone
- Certain trichothecenes
Clay binders are commonly used in livestock feed to reduce mycotoxin absorption and have been investigated as dietary binders for humans in some research settings.
Glucomannan (Yeast-Derived Polysaccharides)
Glucomannan is a soluble fiber derived from konjac root, but in mycotoxin-binding research the term often refers to modified yeast cell wall polysaccharides, particularly glucomannan complexes.
These polysaccharides may bind certain mycotoxins through hydrogen bonding and molecular adsorption. Research suggests glucomannan may interact with:
- Aflatoxins
- Ochratoxin A
- Zearalenone
Yeast cell wall preparations are widely used in agriculture as mycotoxin adsorbents in animal feed, and some functional medicine protocols adapt similar compounds for human use.
Chlorella
Chlorella is a freshwater microalgae rich in chlorophyll, fiber, and bioactive compounds. Some laboratory studies suggest chlorella may interact with environmental toxins through surface binding and bioaccumulation mechanisms.
While most research has focused on heavy metals and organic pollutants, chlorella has been proposed as a supportive binder for certain mycotoxins due to its cell wall structure and polysaccharide content.
Additionally, chlorella provides nutrients and antioxidants that may support general detoxification pathways.
Humic and Fulvic Acids
Humic acids are complex organic molecules formed through the decomposition of plant matter in soil. These substances have strong adsorption capabilities due to their large molecular structure and multiple binding sites.
Humic compounds have demonstrated the ability to bind various toxins, including:
- Mycotoxins
- Heavy metals
- Organic pollutants
Due to their broad binding capacity, humic substances are sometimes used often in integrative detoxification protocols.
Modified Citrus Pectin and Other Polysaccharide Fibers
Fibers such as modified citrus pectin and plant mucilages may also act as mild binding agents within the gastrointestinal tract.
These compounds form gel-like matrices that may:
- Trap toxins within the intestinal lumen
- Promote elimination through stool
- Support beneficial gut microbiota
Although their binding capacity may be weaker than pharmaceutical binders, fiber-based approaches can support long-term gastrointestinal detoxification strategies and provide a gentler solution for children or those with compromised detoxification capabilities.
Chitosan and Other Marine-Derived Binders
Chitosan is derived from chitin found in shellfish exoskeletons and has been studied for its adsorption capacity.
Because of its positively charged structure, chitosan can interact with negatively charged compounds and may help bind certain toxins, including some mycotoxins, in experimental models.
Probiotics and Microbial Detoxification
Certain probiotic species may contribute to mycotoxin detoxification through mechanisms such as:
- Binding toxins to microbial cell walls
- Biotransformation of toxins into less harmful metabolites
- Inhibition of fungal growth or mycotoxin production
- Supporting gut barrier integrity
In laboratory studies, many probiotic effects appear to be driven by binding of mycotoxins to bacterial or yeast cell wall components, such as peptidoglycans, teichoic acids, and polysaccharides. These structural components can interact with toxin molecules and reduce their bioavailability in the gut.
Lactic Acid Bacteria (LAB)
Several Lactobacillus and Bifidobacterium species have demonstrated the ability to bind or reduce certain mycotoxins in experimental models. Among the most studied strains:
- Lactobacillus rhamnosus GG (LGG) – One of the most extensively researched probiotic strains for mycotoxin binding. Studies show it can bind aflatoxin B1, with some experiments demonstrating strong and stable binding capacity across multiple conditions.
- Lactobacillus casei – In vitro research suggests this species can bind aflatoxin B1 and aflatoxin M1, potentially reducing toxin bioavailability.
- Lactobacillus acidophilus – Experimental studies have demonstrated significant binding of aflatoxin B1 in simulated food and storage systems.
- Lactobacillus plantarum – Some strains may bind aflatoxins and help limit the growth of toxin-producing fungi in food systems.
Across studies, the primary mechanism appears to be adsorption of toxins to bacterial cell walls rather than enzymatic degradation, meaning the bound toxin may pass through the gastrointestinal tract and be eliminated in stool.
Saccharomyces boulardii
The probiotic yeast Saccharomyces boulardii has also demonstrated potential mycotoxin-binding activity. Its cell wall contains mannans and β-glucans, which can adsorb mycotoxins and reduce intestinal absorption. Research suggests S. boulardii may bind aflatoxins and ochratoxin A, while also modulating inflammatory signaling pathways associated with toxin exposure.
Some studies suggest that combinations of probiotic organisms—such as S. boulardii with Lactobacillus rhamnosus or Lactobacillus plantarum—may enhance binding capacity in experimental systems.
When integrated into a broader clinical approach (including environmental exposure reduction, nutritional support, and targeted binders), probiotics may contribute to a multi-mechanistic strategy for managing mycotoxin exposure.
N-Acetyl Cysteine (NAC): Supporting Detoxification Rather Than Binding
N-acetyl cysteine (NAC) is not a traditional binding agent. Instead, it supports detoxification through a different mechanism.
NAC serves as a precursor to glutathione, one of the body’s most important endogenous antioxidants. Glutathione plays a central role in phase II liver detoxification, where toxins, including some mycotoxins, are conjugated to increase their water solubility and facilitate elimination through bile or urine.
In experimental research, NAC has also demonstrated the ability to:
- Reduce oxidative stress induced by mycotoxins
- Support cellular antioxidant defenses
- Protect mitochondrial function in toxin exposure models
For these reasons, NAC is often included alongside binder protocols to support biotransformation and antioxidant defense, even though it does not directly bind toxins in the gastrointestinal tract.
Personalizing Binder Strategies
Since different binders interact with toxins through distinct mechanisms, clinicians often tailor protocols based on an individual’s specific mycotoxin exposure profile.
Tools such as the MycoTOX Profile from Mosaic Diagnostics can help identify which mycotoxins are present and may guide more personalized therapeutic strategies.
For example:
- Aflatoxin exposure may respond well to clay-based binders or glucomannan complexes
- Ochratoxin A protocols may incorporate bile-binding strategies
- Trichothecenes may require a broader multi-binder approach
Understanding the specific toxin pattern can help clinicians select targeted interventions rather than relying on a single generalized approach.
Learn more about the MycoTOX Profile and how urinary mycotoxin testing can support personalized exposure assessment.
Summary
Mycotoxin binders represent one potential strategy for supporting toxin elimination in individuals exposed to mold-derived metabolites. These compounds work through a variety of mechanisms—including adsorption, complex formation, and interruption of enterohepatic recirculation—to reduce toxin reabsorption and promote elimination.
Common binder categories include:
- Cholestyramine
- Activated charcoal
- Clay-based binders
- Yeast-derived glucomannan complexes
- Chlorella and humic substances
- Fiber-based binders
- Chitosan and marine-derived compounds
- Probiotics that support microbial detoxification
In addition, supportive nutrients such as N-acetyl cysteine can enhance detoxification pathways by supporting glutathione production and antioxidant defenses.
Because responses to mold exposure can vary widely, testing and individualized care remain essential. Laboratory tools like the Mosaic Diagnostics MycoTOX Profile can help clinicians evaluate potential exposures and develop targeted strategies to support recovery.
Explore the MycoTOX Profile to better understand patterns of mycotoxin exposure and guide personalized clinical decision-making.
References